Medical Device Post Market Surveillance (PMS) in Singapore
Medical Devices Post-Market Surveillance in Singapore
Medical Device PMS Management for HSA
Compliance and Regulatory Confidence - Singapore
In Singapore, post-market surveillance obligations must be carried out in accordance with Health Sciences Authority (HSA) regulations and national compliance requirements. Once a device is registered and placed on the market, manufacturers and their appointed local representatives must continue to fulfill post-market responsibilities under Singapore’s regulatory framework.
Post-Market Surveillance (PMS) in Singapore establishes the processes for maintaining device safety and performance after commercialization. This includes complaint handling, incident evaluation, reporting obligations, and corrective action management in line with HSA requirements.
At Andaman Medical, we oversee these post-market responsibilities on your behalf. Our team works closely with Singapore’s regulatory framework to maintain structured documentation, coordinate required reporting activities, and support ongoing compliance throughout the product lifecycle.
Why Medical Device PMS Matters in Singapore
Post-Market Surveillance (PMS) is a fundamental component of Singapore’s medical device regulatory system once products are available on the market.
Manufacturers and local representatives are expected to maintain complete records, assess reported issues, and fulfill reporting obligations under HSA guidelines.
Here’s how Andaman Medical helps you overcome the challenges of medical device post-market surveillance in Singapore:
Regulatory Compliance
Regulatory Compliance
PMS is a mandatory requirement by the Ministry of Health to ensure the safety, effectiveness, and quality of medical devices. Regular monitoring maintains compliance and avoids penalties. We manage your PMS activities, ensuring full compliance while mitigating risks of non-compliance and penalties.
Early Detection of Issues
Early Detection of Issues
Post-market surveillance identifies potential safety concerns, performance issues, or adverse events early. This proactive approach prevents widespread problems and ensures patient safety. We conduct monitoring and reporting to identify risks, enabling timely corrective actions to protect patients and your product’s reputation.
Improvement of Product Quality
Improvement of Product Quality
Monitoring device performance in real-world settings provides valuable feedback to improve product design, manufacturing, and quality. These enhancements lead to better patient outcomes and stronger market presence. We analyse PMS data to deliver insights that refine your products and maintain high-quality standards.
Maintaining Market Authorisation
Maintaining Market Authorisation
Effective PMS is essential for maintaining your device’s market authorisation. Meeting surveillance requirements demonstrates your commitment to safety and helps avoid losing regulatory approval. We ensure you meet all PMS obligations, keeping your device compliant and authorized in Malaysia.
Building Trust with Healthcare Providers
Building Trust with Healthcare Providers
PMS data demonstrates ongoing device safety and performance, building confidence among healthcare professionals. This trust leads to increased adoption of your products and stronger relationships. We deliver PMS reports and insights that enhance credibility and strengthen trust with healthcare providers.
Risk Management and Mitigation
Risk Management and Mitigation
Post-market surveillance helps identify and address risks, enabling corrective actions to prevent recalls, legal issues, and reputational damage. We manage risks by identifying issues, implementing corrective measures, and protecting your brand.
Adverse Event (AE) reporting requirements in Singapore
| Adverse events | Report within |
|---|---|
| Serious threat to public health | 48 hours |
| Death | 10 days |
| Serious deterioration in state of health | 10 days |
| Possible death or serious injury if the adverse event were to recur | 30 days |
In accordance with Singapore’s post-market surveillance of medical devices, if there is uncertainty about whether the AE is reportable, you should submit a report within the specified time frames. The clock for reporting starts as soon as any personnel in your company, including sales representatives, are made aware of the AE.
FSCA Reporting Requirements
Field Safety Correction Action (FSCA) is any action taken concerning the medical device to reduce the risk of death or the serious deterioration of the health of a user or patient. Possible FSCAs include:
- Product recall
- Product replacement
- Product destruction
- Product modification including any retro-fitting
- Permanent or temporary changes to the labelling or IFU
- Software upgrades
The reporting person should be the dealer that manufactured, imported, supplied or the product registrant for the device concerned. In cases where more than one dealer is involved, each dealer will need to report the FSCA individually.
Before initiating the FSCA, the dealer must notify the HSA. Once it’s been reported, the dealer can proceed without waiting for approval from the HSA.
Medical device dealers must report the Field Safety Corrective Action via the newly launched Online Safety, Compliance Application and Registration (OSCAR) System.
Medical Device PMS Requirements in Singapore
Every manufacturer, importer, supplier, or registrant of a medical device must maintain a record of every received complaint and produce that record for inspection by HSA. The record must contain detailed information as per the Health Products Act (Medical Devices) Regulations, 41.
They are required to plan, establish, document, implement, maintain, and update a PMS system for medical devices placed on the Singapore market. The system must be designed and documented in a PMS plan and then constantly updated according to a structured assessment of how well it is working and the data it has yielded. Post-market surveillance for medical devices is required for all events occurring in Singapore in accordance with the Health Products Act and the Health Products (Medical Devices) Regulation 2010.
The HSA uses a number of post-market surveillance of medical devices and risk assessment measures to ensure the safety of medical devices. These measures include:
- mandatory reporting by medical device dealers (economic operators such as manufacturers and distributors)
- reporting from healthcare professionals
- and an information exchange with medical device regulatory agencies in other ASEAN member states.
The HSA requires that data generated by Singapore’s PMS for medical device system is used in specific ways; in particular it should be used to:
- update the device’s benefit-risk analysis
- feed improvements to risk management
- update the device’s design, Instructions For Use (IFU) and labels
- update the device’s clinical evaluation
- to identify needs for Corrective And Preventive Actions (CAPA) or Field Safety Corrective Actions (FSCA).
The PMS plan should collect data from a range of different sources including:
- Serious incidents
- Field Safety Corrective Actions (FSCAs)
- persons affected by the incident (only for AE)
- Complaints
- Databases and/or registries
- Feedback
In addition to the requirement for manufacturers to implement a PMS for medical devices in Singapore, there are specific requirements for adverse events and FSCAs applicable to all economic operators (manufacturers, importers, distributors, local authorised representatives).
Adverse Event (AE) reporting requirements in Singapore
| Adverse events | Report within |
|---|---|
| Serious threat to public health | 48 hours |
| Death | 10 days |
| Serious deterioration in state of health | 10 days |
| Possible death or serious injury if the adverse event were to recur | 30 days |
In accordance with Singapore’s post-market surveillance of medical devices, if there is uncertainty about whether the AE is reportable, you should submit a report within the specified time frames. The clock for reporting starts as soon as any personnel in your company, including sales representatives, are made aware of the AE.
FSCA Reporting Requirements
Field Safety Correction Action (FSCA) is any action taken concerning the medical device to reduce the risk of death or the serious deterioration of the health of a user or patient. Possible FSCAs include:
- Product recall
- Product replacement
- Product destruction
- Product modification including any retro-fitting
- Permanent or temporary changes to the labelling or IFU
- Software upgrades
The reporting person should be the dealer that manufactured, imported, supplied or the product registrant for the device concerned. In cases where more than one dealer is involved, each dealer will need to report the FSCA individually.
Before initiating the FSCA, the dealer must notify the HSA. Once it’s been reported, the dealer can proceed without waiting for approval from the HSA.
Medical device dealers must report the Field Safety Corrective Action via the newly launched Online Safety, Compliance Application and Registration (OSCAR) System.
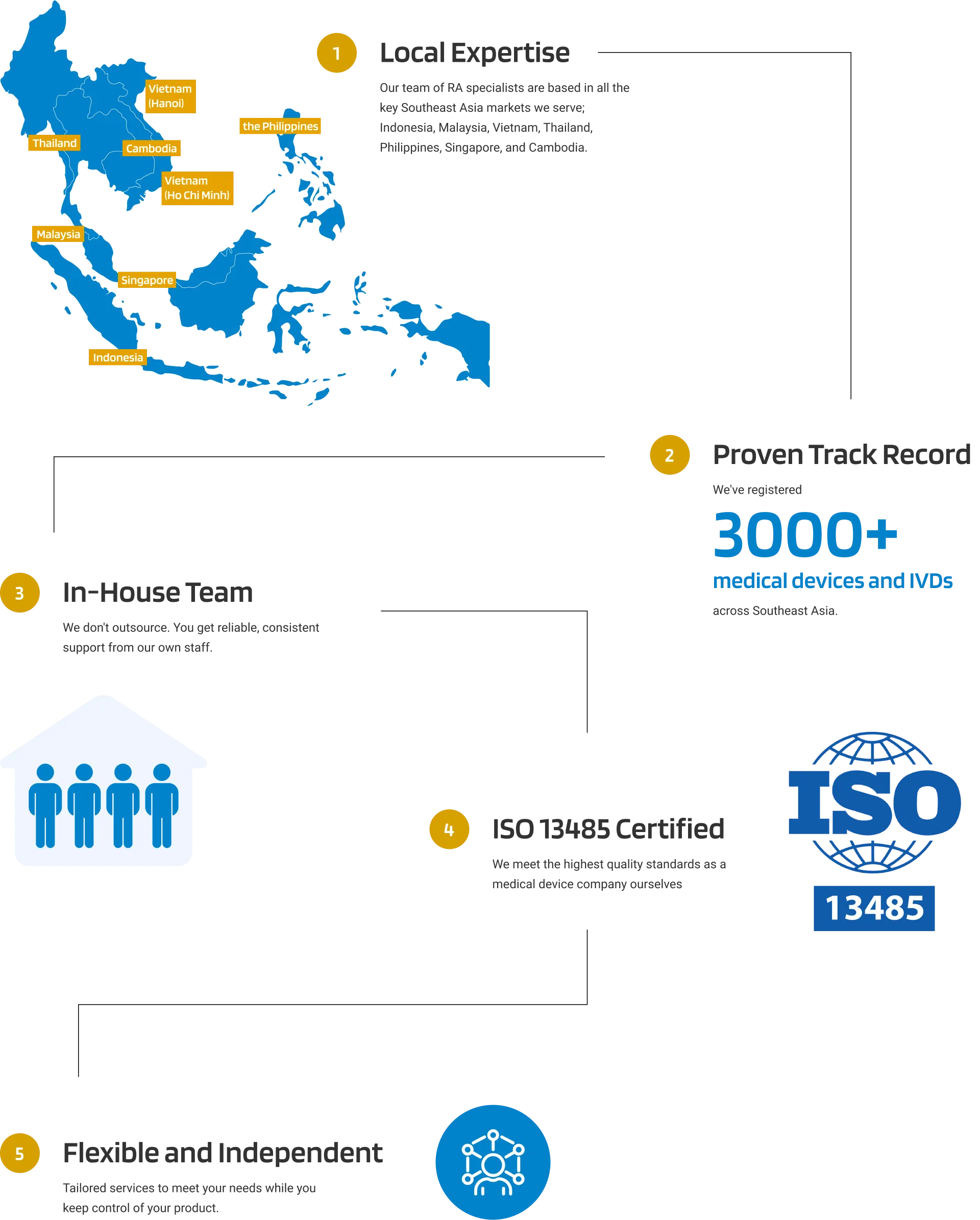
Why Choose Andaman Medical?
Our expert guidance through the region's evolving regulatory landscape ensures your product remains compliant, allowing you to bring your products to market efficiently and effectively.
Experienced In-house Team
across 7 Southeast Asian Countries
Full Control with Seamless
Distributor Switching
Singular Point of Contact
for Registration
QMS Compliance
ISO 13485 Certified
Overcome Language Barrier
With Multilingual Local Team
One-Stop Access
into Southeast Asia