Medical Device Post Market Surveillance (PMS) in Cambodia
Medical Device Post-Market Surveillance in Cambodia
Navigating Cambodia’s regulatory landscape for medical device post-market surveillance can be complex. With Andaman Medical’s expert PMS solutions, we take the burden off your shoulders by managing the entire process for you. From complying with local regulations to monitoring device performance and safety, we ensure your products remain compliant, reliable, and market-ready for long-term success.
Medical Device PMS Support for Regulatory
Compliance and Ongoing Risk Management
- Cambodia
Medical device post-market surveillance in Cambodia requires structured oversight and a clear understanding of national regulatory expectations. After a device is approved and introduced to the market, manufacturers and their appointed local representatives must continue to fulfill post-market obligations under Cambodia’s regulatory framework.
Post-Market Surveillance (PMS) in Cambodia is designed to ensure that medical devices continue to meet established standards for safety, quality, and effectiveness after commercialization. It provides the framework for complaint documentation, incident assessment, regulatory reporting, and corrective action implementation in accordance with local requirements.
At Andaman Medical, we manage these post-market responsibilities on your behalf. Our team supports compliance with Cambodia’s regulatory standards, maintains organized documentation processes, and coordinates required reporting activities to help ensure your devices remain aligned with national expectations.
Why Medical Device PMS Matters in Cambodia
Post-Market Surveillance (PMS) is an important component of Cambodia’s medical device regulatory system once products are available on the market.
Manufacturers and local representatives are responsible for maintaining accurate records, evaluating reported issues, and carrying out required reporting and corrective activities under national guidelines.
Here’s how Andaman Medical helps you navigate the challenges of medical device post-market surveillance in Cambodia:
Regulatory Compliance
Regulatory Compliance
Post-market surveillance (PMS) is a mandatory requirement by the Ministry of Health to ensure continued safety, effectiveness, and quality of medical devices in the market. Regular monitoring helps maintain compliance with local regulations and avoids penalties.
Early Detection of Issues
Early Detection of Issues
PMS allows for the early identification of potential safety concerns, performance issues, or adverse events related to your device. This proactive approach helps prevent widespread problems and protects patients from harm.
Improvement of Product Quality
Improvement of Product Quality
Continuous monitoring provides valuable feedback on device performance in real-world settings. This information can be used to enhance product design, manufacturing processes, and overall quality, leading to better patient outcomes.
Maintaining Market Authorization
Maintaining Market Authorization
Effective post-market surveillance is critical for maintaining your device’s market authorization. Ongoing compliance with surveillance requirements demonstrates your commitment to product safety and helps avoid the risk of losing regulatory approval.
Building Trust with Healthcare Providers
Building Trust with Healthcare Providers
PMS data helps build confidence among healthcare professionals by providing evidence of ongoing device performance and safety. This trust can lead to increased adoption and recommendation of your products.
Risk Management and Mitigation
Risk Management and Mitigation
PMS helps manufacturers identify and address risks associated with their devices, enabling timely corrective actions and reducing the likelihood of costly recalls, legal issues, and damage to brand reputation.
Medical Device PMS Requirements in Cambodia?
Following Annex 5 of the AMDD, the following is required as part of the post-market surveillance (PMS) of medical devices in Cambodia:
Importation and/or Distribution records
Traceability is not only a requirement of an effective quality system but also the requirement of regulatory bodies around the world. Keeping proper and appropriate importation and/or distribution records is an important component of ensuring the traceability and post-market surveillance of medical devices in the Cambodian market.
Adverse events
In Cambodia, several post-marketing risk assessment measures to ensure the continued safe use of medical devices may be undertaken. These measures include reporting from healthcare professionals, mandatory reporting from medical device dealers, and the exchange of regulatory information with other medical device regulatory agencies. The mandatory reporting of AEs by medical device dealers is an important part of the post-market surveillance system for medical devices. The objective of AE reporting and subsequent evaluations is to improve the protection of the health and safety of patients, users, and others by disseminating information that may reduce the likelihood of, or prevent the repetition of AEs, or alleviate the consequences of such repetition.
Complaint records
An effective complaint handling system is an important part of any quality system for medical devices in Cambodia. Any complaint received on a medical device should be evaluated and, if necessary, thoroughly investigated and analysed as part of post-market surveillance. Corrective actions should be taken, and the results of the evaluation should lead to a conclusion regarding whether the complaint was valid, the causes of the complaint, and what actions were necessary to prevent further occurrences.
Field Safety Corrective Action (FSCA)
An FSCA is required when it becomes necessary for the product owner of the medical device to take action (including recall of the medical device) to eliminate or reduce the risk of, the hazards identified. An FSCA may still be necessary even when the medical device is no longer on the market or has been withdrawn but could still possibly be in use (e.g. implants). An FSCA only applies to a medical device that has already been distributed by the product owner. It does not arise when a product owner is exchanging or upgrading medical devices in the absence of a safety risk or when removals from the market are for purely commercial reasons.
Adverse Event Reporting Timeline
| Adverse events | Report within |
|---|---|
| Serious threat to public health | 48 hours |
| Death | 10 days |
| Serious deterioration in state of health | 10 days |
| Possible death or serious injury if the adverse event were to recur | 30 days |
Navigating post-market surveillance in Cambodia can be complex, but Andaman Medical is here to simplify the process and ensure you stay fully compliant. Connect with our experts today and take the first step toward smooth registration!
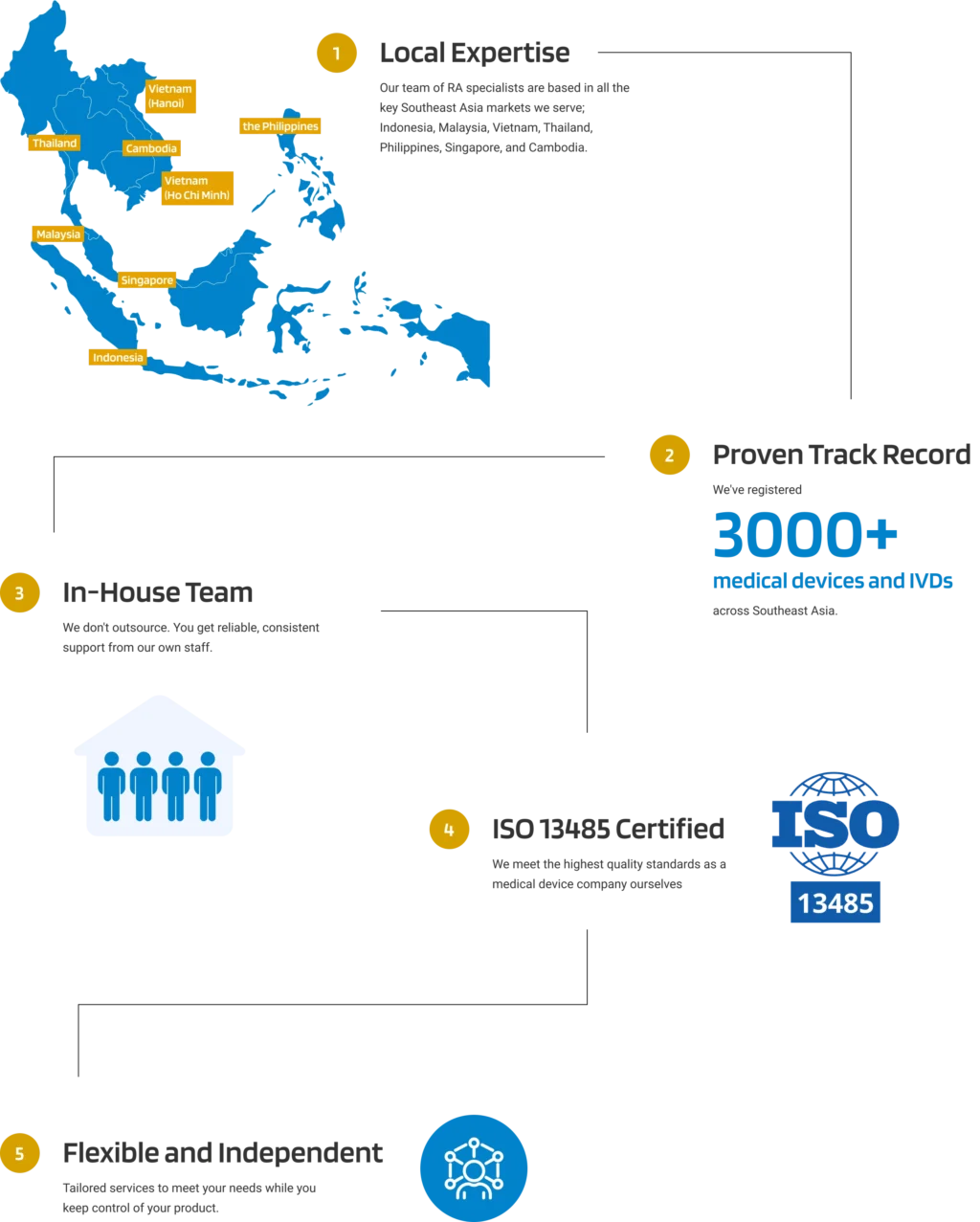
Why Choose Andaman Medical?
Our expert guidance through the region's evolving regulatory landscape ensures your product remains compliant, allowing you to bring your products to market efficiently and effectively.
Experienced In-house Team
across 7 Southeast Asian Countries
Full Control with Seamless
Distributor Switching
Singular Point of Contact
for Registration
QMS Compliance
ISO 13485 Certified
Overcome Language Barrier
With Multilingual Local Team
One-Stop Access
into Southeast Asia
Why Choose Andaman Medical with the Post-Market Surveillance of Medical Devices in Cambodia