Medical Device Post Market Surveillance (PMS) in Thailand
Medical Device Post-Market Surveillance in Thailand
Maintaining compliance, safety, and reliability for your medical devices in Thailand can be complex. That’s where Andaman Medical steps in.
With our expert post-market surveillance services in Thailand, we manage the entire process—from meeting Thai FDA requirements to tracking ongoing product performance and safety. We help ensure your devices remain compliant, trusted, and ready for long-term success in the Thai market.
Medical Device PMS Oversight for Thai FDA
Compliance and Market Continuity - Thailand
Maintaining compliance, safety, and reliability for medical devices in Thailand requires continuous regulatory oversight and familiarity with Thai FDA expectations. After a device is approved and distributed, manufacturers and appointed local representatives must continue to meet post-market obligations under Thailand’s regulatory framework.
Post-Market Surveillance (PMS) in Thailand provides the structure for monitoring device performance after market entry. It includes complaint documentation, incident review, reporting responsibilities, and corrective action management in accordance with Thai FDA requirements.
At Andaman Medical, we manage these post-market responsibilities on your behalf. Our team works closely with Thai regulatory requirements to maintain proper documentation, coordinate reporting activities, and support ongoing compliance throughout the product lifecycle.
Why Medical Device PMS Matters in Thailand
Post-Market Surveillance (PMS) is a critical component of Thailand’s medical device regulatory system once products are available on the market.
Manufacturers and local representatives are expected to maintain complete records, evaluate reported issues, and fulfill regulatory reporting obligations under Thai FDA guidelines.
Here’s how Andaman Medical supports your post-market surveillance needs in Thailand:
Ensuring Patient Safety
Ensuring Patient Safety
- PMS enables manufacturers and regulatory authorities to detect safety issues early by monitoring real-world device performance.
- It helps identify and address potential adverse events that could impact users’ health.
- Timely corrective actions, such as suspending distribution or recalling defective devices, can be taken to protect patients.
Regulatory Compliance
Regulatory Compliance
- Medical device PMS in Vietnam is a regulatory requirement, as outlined in Decree No. 98/2021/ND-CP on Medical Device Management.
- Licence holders must maintain detailed device history records and follow mandatory reporting procedures.
- Non-compliance can result in import delays or loss of market access.
Quality Improvement
Quality Improvement
- PMS data provides valuable insights into device performance, helping manufacturers identify areas for improvement.
- Analysing PMS data supports informed decisions on product enhancements and updates.
- Addressing issues found through PMS can lead to better product quality and reliability.
Market Access and Competitiveness
Market Access and Competitiveness
- A robust PMS system ensures compliance with evolving regulatory requirements.
- Proactive monitoring and resolving of issues give manufacturers a competitive advantage.
- Positive safety and performance data build trust with healthcare providers and patients.
Ongoing Monitoring
Ongoing Monitoring
- The Infrastructure and Medical Device Administration (IMDA) conducts post-market inspections to ensure the quality of imported and circulated devices.
- PMS activities include inspections, sampling, testing, and adverse event reporting.
- Manufacturers and distributors must report any adverse events or product defects to the authorities.
Medical Device PMS Requirements in Thailand
As part of post-market surveillance of medical devices, Thailand requires the reporting of medical device defects or adverse events (AEs) affecting any person, whether the incident occurs within Thailand or abroad, when the medical device is suspected to be a contributory cause. The reporting is required for the following criteria:
- serious threat to public health incidents
- death or serious injury
- if scientific data or evidence indicates that the incident can lead to death or serious harm to consumers if it were to recur.
In order to reduce or eliminate the risks associated with the medical device defect or AE which can result in a serious threat to public health or imminent risk of death or serious harm, the surveillance and reporting of a FSCA is required when the Product Owner undertakes any of the following actions either in Thailand or outside Thailand:
- product recall
- device modification
- product exchange
- device destruction
- safety notification updates and changes.
In order for the Thailand’s FDA to adequately evaluate the cause of the medical device defect or AE, and the subsequent FSCAs, the following information is required as a minimum for the reporting:
- description of the device defect or AE, and FSCA
- place where the device defect or Adverse Effect occurred
- persons affected by the incident (only for AE)
- health hazard evaluation report or any related documents, together with the initial report (only for FSCA).
The Adverse Effect and FSCA reports can be submitted via the online Medical Device Problem Reporting System on the Health Product Vigilance Centre website.
Timelines for AE and FSCA reporting in Thailand
| Type of Report | Timeline | |
|---|---|---|
| 1.1 Adverse Event in Domestic Case | Initial report | Serious threat to public health: Immediately or not later than 48 hours |
| Death or serious injury: Immediately or within 10 days | ||
| Potential to cause death or serious injury if the event recurs: Within 30 days | ||
| Follow-up report | 30 days from initial report | |
| 1.2 Adverse Event in Foreign Case | Report | Twice a year< (1) Incidents occurring from January to June should be reported within the month of August. (2) Incidents occurring from July to December should be reported within the month of February of the following year |
| 1.2. FSCA in Thailand and overseas Domestic Case & Foreign Case | Initial report | Within 48 hours of implementation of the FSCA |
| Follow-up report | Within 21 days of the previous report |
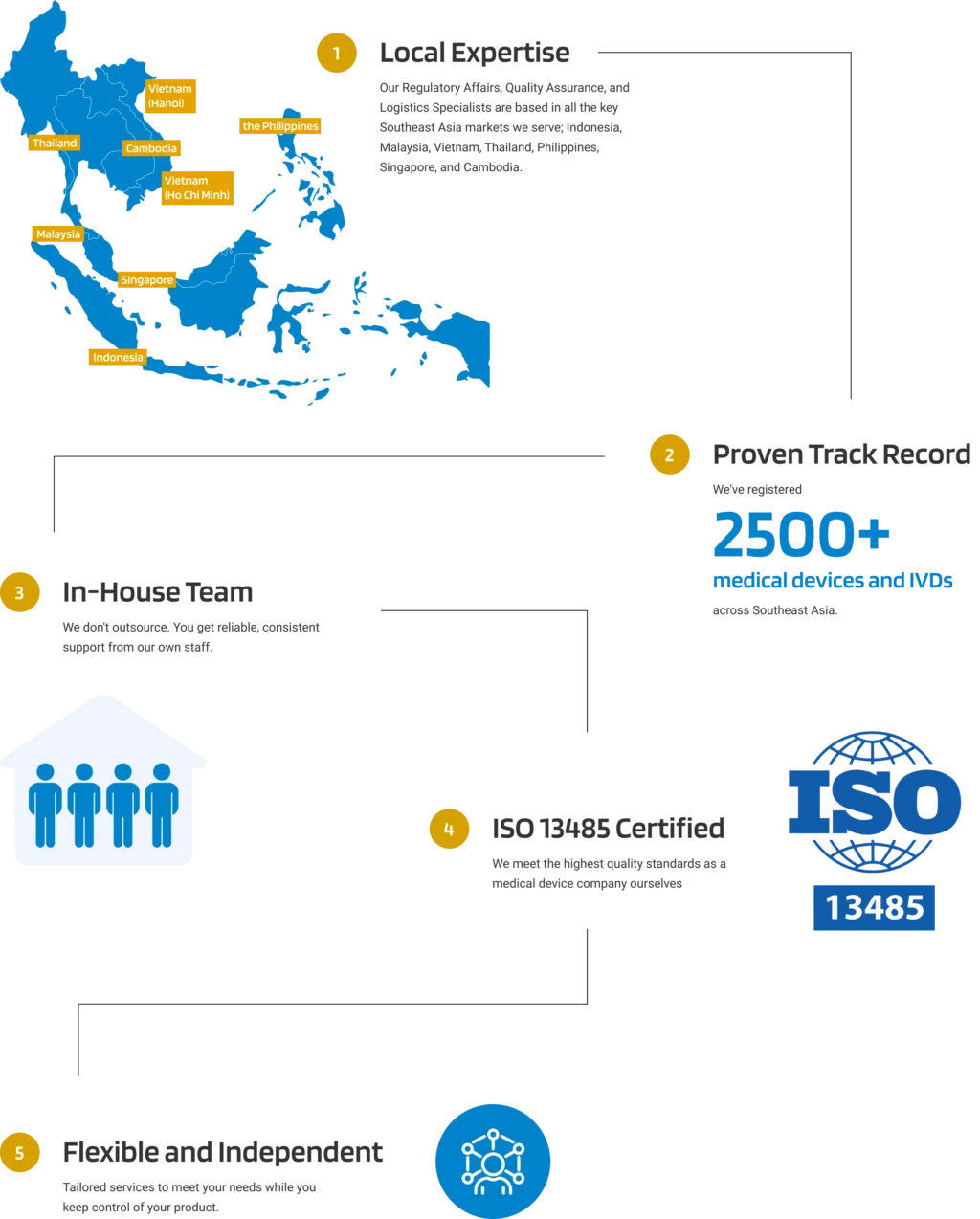
Why Choose Andaman Medical?
Our expert guidance through the region's evolving regulatory landscape ensures your product remains compliant, allowing you to bring your products to market efficiently and effectively.
Experienced In-house Team
across 7 Southeast Asian Countries
Full Control with Seamless
Distributor Switching
Singular Point of Contact
for Registration
QMS Compliance
ISO 13485 Certified
Overcome Language Barrier
With Multilingual Local Team
One-Stop Access
into Southeast Asia
What are the requirements for medical device PMS in Thailand?
Thailand requires the reporting of device defects or AEs happening to any person who is affected by the device either in Thailand or outside Thailand when the medical device is suspected to be a contributory cause of the incident, for the following criteria:
- serious threat to public health incidents
- death or serious injury
- if scientific data or evidence indicates that the incident can lead to death or serious harm to consumers if it were to recur.
In order to reduce or eliminate the risks associated with the device defect or AE which can result in a serious threat to public health or imminent risk of death or serious harm, the reporting of a FSCA is required when the Product Owner undertakes any of the following actions either in Thailand or outside Thailand:
- product recall
- device modification
- product exchange
- device destruction
- safety notification updates and changes.
In order for the Thai FDA to adequately evaluate the cause of the device defect or AE, and the subsequent FSCAs, the following information is required as a minimum for the reporting:
- description of the device defect or AE, and FSCA
- place where the device defect or Adverse Effect occurred
- persons affected by the incident (only for AE)
- health hazard evaluation report or any related documents, together with the initial report (only for FSCA).
The Adverse Effect and FSCA reports can be submitted via the online Medical Device Problem Reporting System on the Health Product Vigilance Center website.
What are the timelines for AE and FSCA reporting in Thailand?
The Thai FDA stipulates timelines for reporting of the incident or FSCA as below:
| Type of Report | Timeline | |
|---|---|---|
| Domestic Case | Initial report | Serious threat to public health: Immediately or not later than 48 hours |
| Death or serious injury: Immediately or within 10 days | ||
| Potential to cause death or serious injury if the event recurs: Within 30 days | ||
| Follow-up report | 30 days from initial report | |
| Foreign Case | Report | Twice a year |
| Domestic Case & Foreign Case | Initial report | Within 48 hours of implementation of the FSCA |
| Follow-up report | Within 21 days of the previous report |
How Andaman Medical can help you with post market surveillance in Thailand
With six local offices throughout Southeast Asia, Andaman Medical provides support for your post-market surveillance activities including adverse effect and FSCA mandatory reporting. Our local, in-house staff liaise with the Thai FDA to help you maintain compliance once your medical device is placed on the market. Our services include:
- support for your mandatory reporting of Adverse Effects and Field Safety Corrective Actions
- identify requirements in medical device directives, standards, and guidance documents to ensure effective implementation of a post-market surveillance system
- evaluate your PMS data to ensure that existing processes and outputs are fully compliant
- monitor and report on any regulatory changes to ensure ongoing compliance